CHAPTER 24 Anesthesia for Pediatric Otorhinolaryngologic Surgery
The art and science of anesthesia must be well integrated in caring for the child undergoing ENT anesthesia. Many procedures, including adenoidectomy, rigid bronchoscopy, and laser removal of papillomas, require short intervals of deep planes of anesthesia and limited movement of the patient. To effectively manage operating room services, the patient must awake in a timely manner without airway irritability. One of the major changes in pediatric anesthesia over the past decade has been the abandonment of halothane in favor of sevoflurane. Although sevoflurane appears to be a safer anesthetic agent than halothane, sevoflurane-induced postoperative delirium continues to challenge the pediatric anesthesiologist (Vlajkovic and Sindjelic, 2007; Kuratani and Oi, 2008).
Propofol has many properties that make it a useful anesthetic agent for ENT surgery. It causes less airway irritability than thiopental. Propofol may be protective against and therapeutic for laryngospasm (Brown et al., 1991; Scanlon et al., 1993; Allsop et al., 1995; Afshan et al., 2002). Propofol also has useful antiemetic properties and a low incidence of emergence delirium (Uezono et al., 2000; Moore et al., 2003; Voepel-Lewis et al., 2003), making this anesthetic a good choice for ENT anesthesia.
Anesthesia for otologic procedures
Otitis media is the most frequently diagnosed childhood malady, with the highest incidence occurring among children between the ages of 6 and 18 months. Its frequency decreases after the first year of life. By age 7 years, otitis media is a less frequent pediatric diagnosis. Otitis media is inflammation of the middle ear, without reference to pathogenesis or cause. The other areas of the temporal bone that are contiguous with the middle ear, including the mastoid, petrous apex, and perilabyrinthine air cells, also may be involved. In those who have had a single episode of acute otitis media, residual middle ear fluid remains in up to 40% of patients at 1 month, 20% at 2 months, and 10% at 3 months (Pelton et al., 1977; Teele et al., 1980; Gluckman, 1990).
Many patients with congenital anomalies are candidates for myringotomy tubes. Patients with cleft palate, craniofacial malformations, Down syndrome, Turner’s syndrome, and human immunodeficiency virus (HIV) infection have a higher incidence of otitis media and therefore have a greater need for myringotomy and insertion of tympanostomy tubes than the general population (Bluestone and Klein, 2003). These children have other airway and medical issues that complicate the usual approach to the healthy child requiring myringotomy tubes. Critically ill patients with a history of prolonged nasotracheal intubation have a high incidence of persistent middle ear effusions requiring middle ear ventilation.
Adenoidectomy for chronic otitis media with effusion may benefit some children. The effectiveness of adenoidectomy for chronic otitis media with effusion is not directly related to adenoid size. For children who have recurrent or chronic otitis media with effusion and who had one or more myringotomy and tympanostomy tube insertion procedure in the past, adenoidectomy may be a reasonable option (Paradise et al., 2003). Upper airway obstruction, recurrent acute or chronic adenoiditis, and sinusitis are additional indications for adenoidectomy in children who have chronic otitis media with effusion.
Myringotomy and Insertion of Tympanostomy Tubes
Candidates for myringotomy and tube insertion present with subacute or chronic upper respiratory tract infection with or without fever. In these children, surgical intervention appears to improve the symptoms of upper respiratory infection postoperatively (Tait and Knight, 1987). However, the finding by these investigators is applicable only to myringotomy for children with middle ear effusion; it should not be generalized or applied to children with upper respiratory infection (especially in its acute phase with fever) who are scheduled for other surgical procedures (Hinkle, 1989).
Preoperative sedation should be administered based on the child’s anxiety level. Mask induction of anesthesia with nitrous oxide, oxygen, and sevoflurane provides effective and smooth induction and maintenance anesthesia with prompt emergence. Preoperative or intraoperative analgesia is advocated for early postoperative pain relief. Because these children are usually anesthetized without intravascular access, preoperative administration of oral acetaminophen or nonsteroidal antiinflammatory drugs (NSAIDs) (Watcha et al., 1992) and intraoperative intranasal fentanyl or butorphanol (Bennie et al., 1998; Galinkin et al., 2000; Finkel et al., 2001) or intramuscular ketorolac (Pappas et al., 2003) has been studied and recommended. It appears that intraoperative ketorolac (0.5 mg/kg, up to 30 mg) administered by the intramuscular route provides better analgesia than oral or rectal acetaminophen. Ketorolac is associated with less vomiting and minimal sedation compared with intranasal butorphanol and perhaps with fentanyl. However, ketorolac, along with other NSAIDs, should be used with caution because ketorolac affects platelet function, and increased postoperative bleeding during tonsillectomy or adenoidectomy has been reported (Dahl and Kehlet, 1994; Nikanne et al., 1997).
Because children undergoing myringotomy commonly have adenoidal hypertrophy, they may exhibit signs of nasal and upper airway obstruction (Bluestone and Klein, 2001). These children often require continuous positive airway pressure (CPAP) during inhalational induction and insertion of an oropharyngeal airway after reaching a surgical plane of anesthesia to maintain a patent airway (see Chapter 3, Respiratory Physiology, and Chapter 13, Induction, Maintenance, and Recovery). Although myringotomy and tube insertion may last less than 10 minutes, it should be performed with standard monitoring (i.e., using a precordial stethoscope, pulse oximeter, electrocardiogram, thermometer, and automated blood pressure cuff). Myringotomy is one of a few exceptions in modern pediatric anesthesia practice in the United States for which intravenous infusion is not routinely administered.
Postoperative hypoxemia occurs frequently in infants and children during transport and in the recovery room, even after minor surgical procedures such as myringotomy (Motoyama and Glazener, 1986). After myringotomy, patients are prone to upper airway obstruction, and these children should be transferred to the recovery room in the lateral position while receiving supplemental oxygen (see Chapter 13, Induction, Maintenance, and Recovery).
Mastoidectomy and Tympanoplasty
The clinical importance of the mastoid process is related to its contiguous structures, which include the posterior and middle cranial fossa, sigmoid and lateral sinuses, facial nerve, semicircular canals, and petrous tip of the temporal bone. The distal part of the middle ear and mastoid are connected by the aditus ad antrum, making the mastoid susceptible to infection by infectious processes from the middle ear. With improved medical and surgical management of otitis media, mastoidectomy for mastoiditis has declined considerably over the past several decades. However, mastoidectomies may also be performed in association with cholesteatoma removal and tympanoplasty. Tympanoplasty is performed in patients with chronic perforation or atelectasis of the tympanic membrane (Bluestone and Klein, 2003). Deep retraction pockets may lead to squamous epithelium proliferation in the middle ear and mastoid cavities, causing a cholesteatoma that may grow by the enzymatic activity of the skin tissues and accumulation of squamous debris.
Tympanoplasty is an operation to reconstruct the tympanic membrane with or without grafting. Its primary indications are repair of tympanic perforation; stabilization or improvement of hearing; removal or prevention of congenital, iatrogenic, or primary or secondary acquired cholesteatoma; and removal of atelectatic or diseased areas of the tympanic membrane (Haynes and Harley, 2002). Grafting materials include fat, fascia, perichondrium, periosteum, cartilage, vein, and paper patch.
Mastoidectomy involves the surgical exposure and removal of mastoid air cells. There are several types of mastoidectomy (Bluestone and Klein, 2003). In a complete simple cortical mastoidectomy, the mastoid air-cell system is removed, but the canal wall is left intact. The operation is performed for acute or chronic mastoid osteitis, and it is frequently part of the surgical procedure advocated by some surgeons for cholesteatoma.
Anesthetic Management
For mastoidectomy and tympanoplasty, the patient is positioned supine, with the head laterally rotated away from the affected side. It is important to carefully rotate the neck of a patient under general anesthesia because of the risk for atlantoaxial rotational subluxation (Brisson et al., 2000).
Induction of anesthesia can entail inhalational or intravenous induction. The anesthesiologist should consider endotracheal intubation without muscle relaxation or with a short-acting muscle relaxant so the facial nerve can be monitored during surgery. The use of nitrous oxide may be contraindicated during and after placement of the tympanic graft, because nitrous oxide accumulates in a closed gas space and increases the ambient pressure; some surgeons believe this tends to lift the graft away from its new site (Koivunen et al., 1996; Doyle and Banks, 2003).
The most common postoperative complication requiring unscheduled admission to the hospital for children who have undergone tympanomastoidectomy is postoperative nausea and vomiting (PONV) (Megerian et al., 2000). PONV has been attributed to surgical stimulation of the vestibular labyrinth, anesthetic techniques, or a combination of the two (Jellish et al., 1995; Dornhoffer and Manning, 2000). Propofol has antiemetic properties (Ved et al., 1996; Erb et al., 2002b). It appears that propofol used for induction and maintenance of anesthesia is superior to isoflurane or sevoflurane in reducing PONV after middle ear surgery (Jellish et al., 1995, 1999; Moore et al., 2003). The use of intravenous dexamethasone during surgery appears to decrease PONV in patients after undergoing tympanomastoid surgery (Liu et al., 2001).
Cochlear Implantation
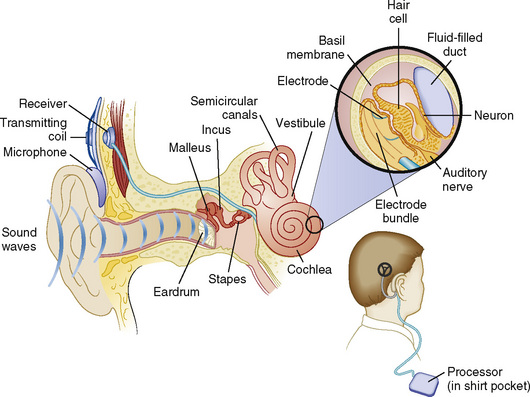
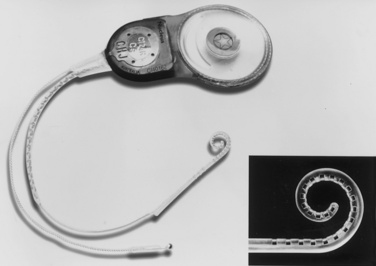
Cochlear implantation has become a feasible choice for profoundly hearing-impaired children (Figs. 24-1 and 24-2). Profoundly deaf children’s auditory nerve fibers remain intact, but the sensory neuroepithelium in the cochlea is absent. This damage occurs because of a genetic defect, infection, cochlear ossification, or aging (Fischetti, 2003). Cochlear implants bypass the damage by receiving and converting sound into signals sent along electrodes to cells adjacent to the auditory nerve. Hearing aids are ineffective because sound cannot be converted into an electrical impulse. Cochlear implants have eight to 22 electrodes that are placed through a cochleostomy. Sounds received from an external microphone are converted to an electrical signal that is received and transmitted by the cochlear nerve (Balkany et al., 2001; Miyamoto and Kirk, 2003). Because the cochlea is full size at birth, there is no anatomic difficulty with electrode insertion in very young children.
Anesthesia for rhinologic procedures
Reduction of Nasal Fracture
A nasal fracture may cause considerable bleeding, and the blood may be swallowed into the stomach. These patients therefore are assumed to have a full stomach, regardless of the time of last food intake. Reduction of a nasal fracture may take only a few moments, but in the acute situation, an oral endotracheal tube is mandatory to protect the airway from pulmonary aspiration. More commonly, surgery is delayed for several days after an isolated nasal fracture to allow swelling to subside. In these cases, a flexible laryngeal mask airway (LMA) can be substituted for an endotracheal tube. The LMA cannot prevent aspiration of stomach contents into the lungs, but it can prevent blood from the nose from passing through the vocal cords (John et al., 1991; Williams and Bailey, 1993). The LMA may be removed either when the patient is deeply anesthetized or when awake.
Nasal Polypectomy
Nasal polypectomy is performed under general anesthesia, often using a preformed Ring, Adair, Ellwyn (RAE) endotracheal tube, which was originally developed by Ring and associates (1975) (see Chapter 10, Equipment). Packing of the pharynx is indicated in most intranasal procedures. One end of the oropharyngeal gauze packing should be tagged with a hemostat and left hanging externally at one corner of the mouth. A note should be prominently placed in the room for all to see, or a labeled tape should be placed across the exit door, reminding all operating room personnel that a throat pack is in place and needs to be removed. Historically, as much as 10 mcg/kg of epinephrine, usually in a 1:200,000 solution (5 mcg/mL), can be used in children for hemostasis under halothane anesthesia without apparent myocardial irritability (Karl et al., 1983; Ueda et al., 1983). More recently, however, our anesthesia practice has decided to use isoflurane, sevoflurane, desflurane, propofol, or remifentanil as maintenance anesthetic, without changing the epinephrine dosage (see Chapter 13, Induction, Maintenance, and Recovery).
Special consideration should be given to the management of children with cystic fibrosis, in whom nasal polyps often occur (Hulka, 2000) (see Chapter 36, Systemic Disorders). The polyps are multiple and recur after removal. These children often have severe obstructive lung disease with recurrent pulmonary infection and thick, tenacious secretions. They are frequently hypoxemic in room air. During general anesthesia, the cystic fibrosis patient may require frequent endotracheal lavage with saline and deep suctioning. Secretions can hamper oxygenation and ventilation; a rise in the peak inspiratory pressure intraoperatively may indicate that the endotracheal tube requires suctioning. An inhaled bronchodilator should be immediately available to treat bronchospasm. Inhaled anesthetics and nitrous oxide or opioids and a relaxant may be used for patients with mild to moderate airway obstruction. Nitrous oxide should be avoided in those with severe lower airway obstruction with air trapping, because of possible hyperinflation of trapped air spaces. These patients with lower airway obstruction should be ventilated with an oxygen-air mixture, and appropriate positive end-expiratory pressure (5 to 7 cm H2O) should be added throughout the procedure to minimize airway closure and atelectasis (see Chapter 3, Respiratory Physiology in Infants and Children). Because of the association of nasal/ethmoidal polyposis with asthma and aspirin sensitivity (Samter’s triad), NSAIDs should be used with caution.
Sinus Surgery
Sinus surgery is indicated for children who have failed maximal medical therapy. Children with allergy or gastroesophageal reflux disease should receive maximal medical therapy and should rarely require surgery (Goldsmith and Rosenfeld, 2003). Children with immune deficiency, immotile cilia syndrome (i.e., Kartagener’s syndrome), or cystic fibrosis (Gysin et al., 2000) are at high risk for chronic sinusitis. Unfortunately, because of their underlying disease, these children have a significant surgical failure rate (Herbert and Bent, 1998).
In children with recurrent sinusitis without chronic disease, adenoidectomy should be the initial procedure if the quantity of adenoid tissue visualized on endoscopy is considered sufficient to serve as a reservoir of bacterial pathogens. Adenoidectomy or adenotonsillectomy is usually the first-line surgical intervention for preschoolers, and it is often appropriate in older children. The expected rate of improvement is 70% to 80% (Goldsmith and Rosenfeld, 2003).
Endoscopic sinus surgery (ESS) should be performed only when children have failed previous therapies. In contrast to older traditional techniques of sinus surgery, ESS focuses on enlarging the natural ostia of the maxillary and ethmoid sinuses, while preserving most or all of the sinus mucosa (Goldsmith and Rosenfeld, 2003). In addition to children with chronic sinusitis who fail medical management, Lusk (2003) lists the accepted indications for ESS as complete nasal obstruction in patients with cystic fibrosis, antrochoanal polyps, intracranial complications, mucoceles and mucopyoceles, orbital abscess, traumatic injury in the optic canal, dacryocystorhinitis resulting from sinusitis, fungal sinusitis, certain neoplasms, and meningoencephalocele. In properly selected children, the results are good, with an expected improvement of 80% (Lusk, 2003). Preoperative computed tomography (CT) is essential in defining the specific diseased sinuses and in looking for anatomic abnormalities that need to be addressed, including septal deviation, concha bullosa cells, obstructing Haller air cells, and abnormal middle turbinates (Goldsmith and Rosenfeld, 2003).
Choanal Atresia
Choanal atresia is a congenital malformation in which no connection exists between the nasal cavity and the aerodigestive tract (Prasad et al., 2002); it has an incidence of 1 in 7000 births. The atresia is bony (30%) or mixed membranous and bony (70%). The existence of purely membranous atresia has come into question (Brown et al., 1996). Choanal atresia is unilateral in 50% to 60% of patients. Syndromes associated with choanal atresia include Apert’s syndrome, DiGeorge syndrome, trisomy 18, Treacher Collins syndrome, camptomelic dysplasia, and CHARGE association (i.e., coloboma, heart defects, atresia choanae, retardation of growth and development, genitourinary problems, and ear anomalies) (Tewfick et al., 1997).
The symptoms caused by choanal atresia depend on whether the obstruction is unilateral or bilateral. Because neonates are obligatory nasal breathers, those with bilateral disease present with acute respiratory distress. Respiratory distress can be attenuated if the mouth is kept open with an oral airway strapped in place or by a large rubber nipple with a large hole cut in it and kept in place with an umbilical tape around the neck (McGovern, 1961; Hengerer and Wein, 2003). For the neonate with bilateral choanal atresia, surgical nasal correction or tracheostomy must be performed within the first few days of life. Other infants may have only unilateral atresia, with minimal symptoms that can go undiagnosed for months or even years. The most common complaint is intractable unilateral anterior nasal discharge.
A number of surgical procedures have been described for the correction of choanal atresia, including endoscopic, transnasal, transseptal, and transpalatal procedures (Holland and McGuirt, 2001). Timing of surgery for bilateral choanal atresia varies and depends on the infant’s ability to adapt to oral breathing and acquire adequate nutrition. Hengerer and Wein (2003) state that some surgeons advocate “a rule of tens” to guide the timing of surgical intervention. The child must reach 10 weeks of age, weigh 10 pounds, and have a hemoglobin level of 10 g/dL. Other surgeons have demonstrated routine success in newborns 48 to 72 hours old and weighing as little as 1900 g (Werkhaven J, 2003, personal communication).
The anesthetic approach depends on the child’s condition. In general, intravenous induction with a muscle relaxant of intermediate duration, endotracheal intubation with an oral RAE tube, and maintenance with an inhaled agent and opioid or propofol and remifentanil infusion suffice. Rarely, algorithms for difficult neonatal intubation must be used for securing the airway in infants with bilateral choanal atresia. After the atresia is surgically corrected, the surgeon is often faced with the problem of restenosis (Pirsig, 1986); however, recent innovations have created optimism that the restenosis rate can be reduced. Newer endoscopic techniques with powered instrumentation have enhanced the safety and efficacy for choanal atresia repair (Prasad et al., 2002). Mitomycin-C, an aminoglycoside and alkylating agent used as an intravenous antineoplastic agent, can be used topically after choanal atresia repair. Topical application inhibits fibroblast growth and migration and granulation tissue formation responsible for restenosis (Holland and McGuirt, 2001). Topical mitomycin has not caused systemic effects and has not contributed to anesthetic complications. Patients with unilateral obstruction usually do well in the postoperative period and require no special monitoring. However, infants undergoing bilateral repair can exhibit partial or intermittent upper airway obstruction that persists for some time. The infant should be observed closely in the ICU with appropriate monitoring until breathing dynamics have normalized.
Anesthesia for pharyngeal and laryngeal procedures
Tonsillectomy and Adenoidectomy
Tonsillectomy and adenoidectomy (perhaps with the exception of myringotomies) are the most common pediatric surgical procedures performed in the United States. Although tonsillectomy and adenoidectomy are frequently performed procedures, the benefits in relation to cost and risk are still hotly debated (Paradise, 2003). Just about every indication has its advocates and detractors (Bluestone, 2001; Paradise, 2003).
Nocturnal upper airway obstruction, with or without obstructive sleep apnea (OSA), is a common indication for adenotonsillectomy. A mild, partial obstruction in otherwise healthy children is exacerbated by conditions such as achondroplasia, Down syndrome, mucopolysaccharidosis, and obesity. Adenotonsillectomy is considered curative when adenotonsillar hypertrophy is the primary cause of childhood sleep-related breathing disorders (Schechter, 2002). Indications for tonsillectomy include recurrent pharyngotonsillitis, chronic tonsillitis, hemorrhagic tonsillitis, peritonsillar abscess, streptococcal carriage, dysphagia, abnormal dentofacial growth, halitosis, and suspicion of malignant disease (i.e., tonsil asymmetry) (Darrow and Siemans, 2002). Additional indications for adenoidectomy include recurrent or chronic rhinosinusitis or adenoiditis, and recurrent otitis media (Darrow and Siemans, 2002). Adenotonsillar hypertrophy with resultant upper airway obstruction is the most frequent cause of OSA in children (Fig. 24-3).
Obstructive Sleep Apnea Syndrome
Obstructive sleep apnea syndrome (OSAS) is a disorder of breathing during sleep characterized by prolonged partial upper airway obstruction (obstructive hypopnea) or intermittent complete obstruction (OSA) with or without snoring and associated with moderate to severe oxygen desaturation that disrupts normal sleep-time breathing and normal sleep patterns (American Thoracic Society, 1996). The most common cause of OSAS among children is upper airway narrowing with adenotonsillar hypertrophy. OSAS also occurs in infants and children with upper airway narrowing resulting from craniofacial anomalies, and in those with neuromuscular diseases, including cerebral palsy and muscular dystrophy (Marcus, 2001; Schwengel et al., 2009) (Box 24-1). See related video online at www.expertconsult.com.![]()
Box 24-1 Some Congenital and Medical Conditions Associated with Obstructive Sleep Apnea Syndrome
From Sterni LM, Tunkel DE: Obstructive sleep apnea in children: an update, Pediatr Clin North Am 50:427, 2003.
In recent years, the epidemic increase in the prevalence of obesity during childhood seems to be contributing to substantial changes in the cross-sectional demographic and anthropometric characteristics of the children being referred for evaluation of OSAS. Although less than 15% of all symptomatic habitually snoring children were obese (i.e., >95th percentile for age and gender) in the early 1990s, more than 50% fulfilled the criteria for obesity among all referrals to a Kentucky sleep center by the mid 2000s (Gozal et al., 2006; Schwengel et al., 2009).
Adenotonsillar hypertrophy is the most common cause of OSAS in children. Many published papers, primarily case reports and case series, support the idea that tonsillectomy with or without adenoidectomy is often the cure for OSAS (Schechter et al., 2002; Garetz, 2008).
The peak prevalence of OSAS in children occurs between 2 and 8 years, which is the age when the tonsils and adenoids are large in relation to the child’s upper airways. The site of collapse is most commonly at the level of the adenoids or velopharynx (Isono et al., 1998; Isono, 2006). Although OSAS is associated with adenotonsillar hypertrophy, there must be other neuromuscular factors involved. These patients usually do not obstruct while awake, implying that sleep induces another dimension to OSAS. Some otherwise normal children with OSAS but with smaller tonsils and adenoids are cured by adenotonsillectomy, whereas others with larger tonsils and adenoids are not (Marcus, 2001). The patency of pharyngeal airway is maintained by tonic and phasic contractions of the upper airway dilator muscles, such as the genioglossus, geniohyoid, and velopalatine muscles (Isono, 2006). Compared with other inspiratory muscles (i.e., diaphragm and intercostal muscles), these upper airway muscles are preferentially depressed with sleep, sedatives, and general anesthetics (Ochiai et al., 1989, 1992) (see Chapter 3, Respiratory Physiology in Infants and Children). It is hypothesized that children with OSAS may have abnormal centrally mediated activation of their airway muscles, leading to a more collapsible upper airway (Marcus et al., 1994).
Fatty deposits of the upper airway structures, and subcutaneous fat deposits in the anterior neck region are the direct causes of airway obstruction in obese children (mostly adolescents), as well as in obese adults, with OSAS. In addition, increased adipose deposits in the chest wall and abdomen can reduce chest wall compliance as well as cephalad displacement of the diaphragm, causing inefficient ventilation by mass loading and decreased functional residual capacity of the lung (Dayyat et al., 2007; Schwengel et al., 2009). Obese patients have symptoms that differ from those in children with OSAS caused by adenotonsillar hypertrophy. Obese patients tend to have a significantly greater incidence of daytime sleepiness, insulin resistance, and systemic as well as pulmonary hypertension than children with adenotonsillar hypertrophy (Tauman et al., 2004). Furthermore, Tauman and coworkers recently observed a high failure rate for obese children with OSAS undergoing adentotonsillectomy.
Dayyat and colleagues (2007) differentiated between children with OSAS into two types. The type 1 child with OSAS is not obese but has disordered breathing secondary to marked lymphadenoid hypertrophy. The type 2 patient is obese with minimal lymphadenoid hyperplasia. Table 24-1 compares the common symptoms experienced by the two types of patients (Dayyat et al., 2007).
TABLE 24-1 Clinical Presentation of Pediatric Obstructive Sleep Apnea Types I and II
| Symptoms and Findings Seen with Similar Frequencies in Types I and II |
| Symptoms and Findings that Differ Between Types | Type I | Type II |
| Excessive daytime sleepiness | + | ++++ |
| Weight gain | − | ++ |
| Hyperactive behavior | ++++ | − or + |
| Truncal obesity | − or + | +++ |
| Enlarged neck circumference | − or + | +++ |
| Enlarged tonsils or adenoids | ++++ | ++ |
| Depression and low self-esteem | + | +++ |
| Shyness and social withdrawal | + | +++ |
| Left ventricular hypertrophy | ++ | ++++ |
| Systemic hypertension | + | ++++ |
| Recurrent ear infections | +++ | − or + |
| Insulin resistance | − | ++++ |
| Dyslipidemia | + | ++++ |
| Elevated C-reactive protein | ++ | ++++ |
| Elevated liver enzymes | − | ++ |
−, Absent; + infrequent to ++++ very frequent.
From Dayyat E, Kheirandish-Gozal L, Gozal D: Childhood obstructive sleep apnea: one or two distinct disease entities? Sleep Med Clin 2:433, 2007.
One of the most interesting areas of childhood OSAS research has been on the effects that OSAS has on systemic inflammation. Children with OSAS are at risk for developing inflammatory responses that may lead to endothelial dysfunction and atherogenesis (Gozal et al., 2008). School-aged children with OSAS have a higher serum C-reactive protein level than controls, and the elevation correlates with the severity of OSAS, and levels decrease after adenotonsillectomy (Gozal et al., 2007). Serum proteomic patterns in children with OSAS differ significantly from those in children who have primary snoring but do not have OSAS (Shah et al., 2006). In the Shah group’s study, proteomic profiling of serum samples in children with OSAS revealed differential expression of circulating proteins. Proteomics may play a future role in diagnosing OSAS in the snoring child.
Symptoms of OSAS include nocturnal snoring, breathing pauses, gasping, use of accessory muscles of respiration, enuresis, and excessive sweating (Messner, 2003). In addition, children with OSAS have a host of sequelae, which are usually reversible after adenotonsillectomy but can lead to perioperative complications during and after surgery. Children with OSAS may present with failure to thrive, although the cause of this growth failure is unclear (Sterni and Tunkel, 2003). Many of these children experience a growth spurt after surgery. Children with OSAS may have neurocognitive deficits such as poor learning, behavioral problems, and lower grades in school than non-OSAS children. Adenotonsillectomy improves functioning in these children (Marcus, 2001). Recent studies demonstrate that despite negative polysomnographic findings, the benefits of adenotonsillectomy are still observed in children with symptomatic airway obstruction, including improvements in behavior and quality of life (Leong and Davis 2007).
The diagnosis of OSAS is based on a thorough history and physical examination along with appropriate sleep studies, including polysomnography (Table 24-2 and Boxes 24-2 and 24-3). Snoring, increased respiratory efforts, periodic obstructive apnea, and oxygen desaturation while sleeping are the universal features of OSAS, which must be differentiated from the benign condition referred to as primary snoring.
TABLE 24-2 Polysomnographic Characteristics of Obstructive Sleep Apnea in Children and Adults
| Characteristic | Adults | Children |
| Apnea duration | 10 sec | 2 breaths |
| Hypopnea desaturation | ≥ 4% | ≥ 3% |
| Hypopnea duration | 10 sec | 2 breaths |
| Hypopnea nasal pressure drop | > 30% | ≥ 50% |
| Cortical arousals | Common | Uncommon |
| Normal apnea-hypopnea index | <5 | <1 |
From Karlson KH Jr: What’s new in pediatric obstructive sleep apnea? Clin Pulm Med 15:226, 2008.
Box 24-2 Key Questions to Ask Parents
Polysomnography
Pediatric polysomnography includes measurement of end-tidal CO2, electroencephalogram, chin EMG, chest wall movement, airflow through the nose and mouth, leg movement, and oxygen saturation via pulse oximetry (Box 24-4). After all events are reviewed and scored, several indices can be calculated. These include the apnea index and the apnea-hypopnea index, which are calculated for the entire sleep period, for both rapid eye movement sleep and non–rapid eye movement sleep. The term index refers to the number of events divided by the number of hours of sleep (Table 24-3). This calculation allows the comparison of polysomnograms of varying time lengths. The apnea index is determined using only apneas, whereas the apnea-hypopnea index includes both apneas and hypopneas (Wagner and Torrez, 2007).
Box 24-4 Components of Polysomnography*
From American Thoracic Society: Standards and indications for cardiopulmonary sleep studies in children, Am J Respir Crit Care Med 153:866, 1996.
TABLE 24-3 Respiratory Events that Can Be Seen During Polysomnography
| Event | Definition |
| Central apnea | Pause in airflow with absent respiratory effort, scored when >20 sec or 2 missed breaths and a >3% drop in oxygen saturation |
| Obstructive apnea | >90% reduction of airflow despite continuing respiratory effort, scored when event lasts at least 2 missed breaths in children |
| Obstructive hypopnea | >50% reduction of airflow with associated respiratory effort, scored when at least 2 missed breaths and >3% drop in oxygen saturation or arousal |
| Mixed apneas | ≥ 90% reduction in airflow, lasting at least 2 missed breaths, and containing absent respiratory effort initially (a central apneic pause), followed by resumption of respiratory effort without a resumption of airflow (an obstructive apnea) |
| Obstructive hypoventilation | End-tidal CO2 > 50 mm Hg for >25% of the total sleep time, with paradoxical respirations, snoring, and no baseline lung disease |
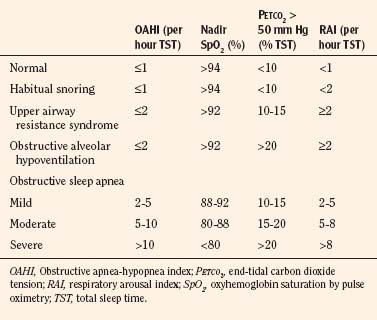
It is helpful to divide the scores into severity-based categories (Table 24-4). OSAS severity can be used to determine the appropriate surgical center where the surgery should be performed and whether the child requires overnight observation (Box 24-5).
Data from McGowan et al., 1992; Gerber et al., 1996; Blum et al., 2004; Fricke et al., 2006; Guilleminault et al., 2007.
Although definitive diagnosis of OSAS is made by a positive polysomnogram, many children are not tested because the polysomnography laboratories specializing in children are few, and the tests are expensive and require an overnight stay (Sterni and Tunkel, 2003). Most children are diagnosed by recommendations set forth by the section of Pediatric Pulmonology Subcommittee on Obstructive Sleep Apnea, sans Polysomnography. This committee’s recommendations for otherwise healthy children older than 1 year are as follows (Schechter, 2002):
Severe, untreated OSAS may lead to pulmonary hypertension and cor pulmonale caused by nocturnal hypoxia and hypercarbia, resulting in compensatory changes in the pulmonary vasculature. Pulmonary vascular resistance increases, causing increased right ventricular strain. Severe cases may progress to pulmonary hypertension, arrhythmias, and cor pulmonale, which are reversible by performing early adenotonsillectomy (Miman et al., 2000). Children with OSAS also tend to have higher diastolic blood pressures. The cardiovascular changes appear to be the result of an increase in sympathetic tone that results from obstructive respiratory events (Marcus et al., 1998). Fortunately, few children develop clinically significant heart failure. It is prudent to pursue an aggressive cardiac evaluation if the child has a loud second heart sound, exercise intolerance, or diastolic hypertension. Because children are diagnosed earlier today, significant heart disease is rarely an issue (Marcus, 2001).
Preoperative Preparation
A careful review of the history, laboratory data, and physical examination results are essential for the optimal outcome of adenotonsillectomy. Careful evaluation of coagulation status is important before performing this procedure. If there is a history of easy bruising, frequent epistaxis, or positive family history, the prothrombin time, partial thromboplastin time, and platelet count should be obtained to rule out coagulopathies. In many pediatric institutions, a coagulation profile is obtained routinely for patients scheduled for adenotonsillectomy. However, in a prospective study of hemostatic assessment of patients before tonsillectomy, routine measurements of a coagulation profile were not useful predictors of postoperative bleeding (Close et al., 1994).
Although relatively mild, von Willebrand’s disease (i.e., reduced factor VIII, decreased platelet adhesiveness because of deficient von Willebrand factor) is the most common coagulopathy seen among patients scheduled for adenotonsillectomy. Children with von Willebrand’s disease or mild hemophilia A are treated preoperatively with desmopressin (1-desamino-8- d-arginine vasopressin [DDAVP]) (Prinsley et al., 1993). DDAVP is a synthetic analogue of vasopressin, which, in addition to its antidiuretic effect, stimulates endothelial cells and releases stored factor VIII and von Willebrand factor (Mannucci, 1988). The intravenous dosage of DDAVP is 0.3 mcg/kg, given over 20 minutes before anesthetic induction.
The examiner should elicit a history of drug ingestion, especially acetylsalicylic acid. If the patient was given such drugs recently, surgery should be postponed, because these drugs cause platelet dysfunction for as long as 10 days and may cause excessive bleeding intraoperatively and postoperatively (Davies and Steward, 1977; Paradise, 2003).
Anesthetic Management
Children who are anxious preoperatively may receive sedation with oral midazolam. In older children scheduled for intravenous induction, EMLA cream (i.e., eutectic mixture of local anesthetics, 2.5% lidocaine and 2.5% prilocaine) is applied to the dorsum of both hands and sealed with plastic adhesives at least 1 hour before intravenous catheter insertion (see Chapter 9, Preoperative Preparation, and Chapter 13, Induction, Maintenance, and Recovery).
Adenoidectomy is a relatively short procedure (15 to 45 minutes), and many centers perform anesthesia without neuromuscular blockade and with less opioid than suggested in the following descriptions. Anesthesia is induced most commonly with oxygen, nitrous oxide, and sevoflurane. As with those undergoing myringotomies, children with adenotonsillar hypertrophy often have partial or complete nasal airway obstruction. The mouth must be kept open during the induction of anesthesia until the gag reflex is abolished and the oral airway is inserted. If necessary, moderate CPAP (10 to 15 cm H2O), with jaw thrust during induction (before the patient is deep enough for oral airway insertion) helps to prevent the pharyngeal airway collapse that results from the relaxation of upper airway muscle tone (Reber et al., 2001; Bruppacher et al., 2003).
After the intravenous route is established, atropine or glycopyrrolate may be given for its anticholinergic effects. The patient’s trachea is intubated with a preformed oral (RAE) tube under deep inhalational anesthesia supplemented with a topical lidocaine spray to the vocal cords under a direct vision, an intravenous bolus of propofol, or an intermediate-acting, nondepolarizing muscle relaxant (e.g., cisatracurium, vecuronium). In older children, anesthesia may be induced intravenously with propofol (2 to 3 mg/kg) mixed with lidocaine (1 to 2 mg/mL of propofol) to reduce pain at the injection site, with or without anticholinergic agent or a muscle relaxant. Inhalation anesthetic or propofol infusion is continued thereafter with spontaneous or controlled ventilation. A cuffed endotracheal tube is recommended to reduce the chance of aspiration of blood and secretions and to reduce gas leaks around the tube. A cuffed endotracheal tube, 0.5 to 1.0 mm (inner diameter) smaller than the age-appropriate size, should be chosen to accommodate the passage of the cuff through the subglottis (Khine et al., 1997; James, 2001; Fine and Borland, 2004). The endotracheal tube is immobilized with adhesive tape over the middle of the lower lip (Fig. 24-4). The breath sounds on both sides of the chest should be auscultated carefully to avoid endobronchial intubation. The endotracheal tube is then held in place by the groove-bladed tongue depressor that is a part of the Ring adaptation of the Brown-Davis mouth gag (Fig. 24-5).
Anesthesia is maintained with supplemental opioids to reduce the requirement of the anesthesia maintenance agent and to provide postoperative analgesia. The physician may start with a loading dose of 1 to 2 mcg/kg of fentanyl, 50 to 100 mcg/kg of morphine, or 10 to 20 mcg/kg of hydromorphone given intravenously, with additional doses administered as needed. Children with moderate and severe OSAS should receive lower dosages of opioids to minimize the risk for prolonged apnea during emergence and postoperative upper airway obstruction. Brown and coworkers (2004) have shown that young age and low preoperative oxygen saturation were associated with lower postoperative morphine requirement. It appears that children with a minimal oxygen saturation of 85% or less during preoperative sleep required half the postoperative analgesic dosage required by children whose saturations were 85% or greater (Brown et al., 2006).
The use of NSAIDs during or after tonsillectomy is controversial. A meta-analysis suggested that there is an increased risk for bleeding and increased return to the operating room when NSAIDs such as ketorolac are used (Marret, 2003). Criticism of this meta-analysis focuses on the small number of patients evaluated, the varying types and dosages of NSAID, and the varying surgical techniques used for the tonsillectomy. A Cochrane database review evaluated the role of NSAIDs in posttonsillectomy bleeding and found no significant correlation between NSAID use and increased risk for posttonsillectomy bleeding (Cardwell et al., 2005). Practice among pediatric anesthesiologists varies. Some centers do not use NSAIDs during adenoidectomy or tonsillectomy, whereas others use them routinely (Allford, 2009).
There is evidence that high dosages of dexamethasone (up to 1 mg/kg, 25 mg maximum) reduce postoperative swelling and pain and decrease the incidence of PONV without apparent adverse effects attributable to dexamethasone (Pappas et al., 1998). Children receiving dexamethasone are more likely to advance to a soft-solid diet on the first postoperative day (Steward et al., 2003).
Bleeding during tonsil and adenoid surgery is seldom excessive, but massive hemorrhage has occurred with tearing of the carotid vessels (Smith, 1972, 1980). Homeostasis is usually obtained using electrocautery. There have been several reports of fires due to electrocautery-induced ignition of the endotracheal tube or packing during tonsillectomy. Fires are caused by combustible material in an oxygen-rich environment (Mattucci and Militana, 2003); management of airway fires is discussed later.
Children with OSAS tend to emerge from anesthesia more slowly than children without OSAS. This may be explained by their deficit in sleep arousal mechanisms. They seem to have elevated sleep arousal mechanisms in response to hypercarbia and increased upper airway obstruction (Marcus et al., 1998, 1999). Other subtle disturbances of sleep architecture may also be present (Bandla et al., 1999).
Techniques of Tonsillectomy and Adenoidectomy
The adenoidectomy is performed either with a mechanical or a thermal technique (Hesham, 2009). Mechanical removal of the adenoids may be performed using curettes, adenotomes, or a micro-debrider. These mechanical techniques require nasopharyngeal packing to prevent bleeding, and the surgeon must remember to remove the packing at the end of the procedure. Thermal removal of adenoid tissue may employ electrosurgical cautery, radiofrequency ablation, or lasers, and it usually results in little bleeding.
Mechanical removal of the tonsil has many technical variations. In general, an incision is made in the anterior pillar mucosa to expose the tonsil capsule, and using sharp or blunt dissection, the tonsil and capsule are removed together. Alternatively, a guillotine, or snare, is occasionally used to remove the tonsil without making an incision. This results in exposure of the underlying pharyngeal musculature with the perforating vascular supply to the tonsil, and hemostasis must be established. At the turn of the previous century, snare tonsillectomy with ether anesthesia was the standard, and hemostasis was achieved after prolonged observation of the surgical field to ensure passive wound clotting. This was less than ideal, and the mortality rate was unacceptably high. Today, active hemorrhage control is performed after removal of the tonsils. Some surgeons use dissolvable suture to ligate the bleeding points, but this is becoming less common. More often, electrosurgical cautery is used for hemostasis. This technique completes an electrical arc between the patient and the surgical instrument and has the potential to ignite flammable gases and combustible materials (such as the sponges in the nasopharynx used for tamponade of the adenoid bleeding). Case reports of airway fires have been reported with the use of electrosurgical cautery in tonsillectomy (Mattucci and Militana, 2003). It is suggested that the inspired oxygen levels be maintained as they are for the use of lasers in the airway (see Laser Surgery of the Larynx, later).
Thermal removal of the tonsil uses the electrosurgical unit or a radiofrequency unit for tonsil dissection. The most common radiofrequency unit in use is Coblation (ArthroCare, Austin, TX). Thermal removal of the tonsils and adenoids generally results in less blood loss, but it may be accompanied by slightly more postoperative pain than other techniques. A cuffed endotracheal tube, using moist throat packing and inspired oxygen levels of less than 40%, will minimize the risk for an airway fire (Johnson et al., 2002).
Intracapsular tonsillectomy is a newer technique that uses either the micro-debrider or the radiofrequency ablator to remove the tonsil tissue while preserving the tonsil capsule (Koltai et al., 2002). In effect, this may be considered a selective removal of the tonsillar mass, which is believed to be the most common cause of OSAS. Preservation of the capsule protects the underlying pharyngeal musculature, resulting in less postoperative pain and hemorrhage. Because the vascular supply is interrupted at a more terminal point, the muscular bed can contract around the vessels, providing better hemostasis. Initially, this technique was recommended only for those patients with OSAS. Recurrent infection is considered a contraindication, because this technique leaves behind residual tonsil tissue that may become reinfected. This technique is especially appropriate in those OSAS children 3 years of age and younger because of the lower postoperative pain scores and thus less opioid need (Solares et al., 2005; Colen et al., 2008). In general, the only risk of an intracapsular tonsillectomy is the rare (<1 in 1500) need to return for a complete tonsillectomy because of tissue regrowth leading to recurrent infectious tonsillitis.
At the conclusion of surgery, the surgeon usually suctions the pharynx and larynx under direct vision to prevent bleeding caused by agitation of raw mucosal surfaces by a suction catheter. Suctioning of the stomach contents may also remove swallowed secretions, blood, or residual preoperative sedative medications. Removal of these preoperative medications may decrease excess sedation in the postoperative period. Some surgeons elect to infiltrate 0.25% bupivacaine or other local anesthetic medications into the tonsillar fossa to aid in postoperative pain relief (Naja et al., 2005), whereas others feel that this analgesic technique may lead to premature discharge of patients who will then experience rebound pain when the local anesthetic wears off. This practice has occasionally been associated with airway obstruction after extubation.
Laryngospasm may occur when the patient is extubated after tonsillectomy. Methods for avoiding this problem include extubating the trachea while the child is deeply anesthetized (not recommended for children with OSAS) or almost completely awake (see Chapter 13, Induction, Maintenance, and Recovery). Although intravenous lidocaine has not consistently proved helpful in preventing laryngospasm, application of local anesthetic before intubation or manipulation of the airway can reduce the incidence of laryngospasm during intubation and after extubation (Leicht et al., 1985; McCulloch et al., 1992; Landsman, 1997). It appears that lidocaine deposited locally in the laryngeal area suppresses laryngeal mucosa neuroreceptor transmission. However, lidocaine’s duration of action at laryngeal receptor sites is only 30 minutes, and its administration before intubation may not protect against laryngospasm at the time of extubation (Warner, 1996).
Before extubation, the lungs should be well expanded with an oxygen-air mixture at sustained high positive pressure (35 to 40 cm H2O) several times (the vital capacity maneuver), to reopen intrathoracic airways and reverse atelectasis, which frequently develops during anesthesia and surgery; the patient should then be extubated under positive pressure (Benoit et al., 2002; Tusman et al., 2003) to prevent postoperative oxygen desaturation. Positive airway pressure at the moment of extubation causes a coughing motion, which helps to expel secretions around the vocal cords. Positive extending pressure on the upper airway walls also attenuates the excitation of the superior laryngeal nerve and may diminish the risk for laryngospasm (Suzuki and Sasaki, 1977; Sasaki, 1979) (see Chapter 3, Respiratory Physiology in Infants and Children, and Chapter 13, Induction, Maintenance, and Recovery).
Postoperative Management
Children with hypertrophic tonsils and adenoids tend to have increased airway obstruction in the immediate postoperative period. The presence of blood and secretions in the pharynx and larynx may provoke upper airway reflexes, leading to laryngospasm. These patients tend to become hypoxemic more often and perhaps more severely during the first several hours after surgery than patients undergoing procedures not involving the upper airways (Motoyama and Glazener, 1986). However, they seem to maintain their preoperative levels of oxygen saturation in room air thereafter, as determined with pulse oximetry (Motoyama and Borland, unpublished observations). Fortunately, serious complications after adenotonsillectomy are infrequent. Postoperative hemorrhage, however, does occur and can become a life-threatening catastrophe (discussed later). Postoperative emesis is relatively common because of pharyngeal mucosal irritation from surgery, which may stimulate the glossopharyngeal nerves, or from bloody secretions that are swallowed. It is therefore prudent for the anesthesiologist to administer antiemetics (e.g., ondansetron or metoclopramide) prophylactically, in addition to dexamethasone before the end of surgery (Pappas et al., 1998). Pain after tonsillectomy is mild to moderate and may be controlled by opioids given intraoperatively as part of anesthetic management or by supplementation (morphine, 50 mcg/kg; fentanyl, 0.5 to 1.0 mcg/kg; hydromorphone, 10 mcg/kg) in the postanesthetic care unit. Postoperative pain after adenoidectomy is relatively mild. Acetaminophen is a good adjunct to pain control in these patients.
It has been reported that children with OSAS have reduced opioid requirements after adenotonsillectomy. In addition, children with OSAS spontaneously breathing during halothane anesthesia are more likely to become apneic than non-OSAS children when treated with the same dosage of fentanyl (Waters et al., 2002). However, others have found no morbidity from opioid or benzodiazepine treatment in OSAS pediatric patients undergoing adenotonsillectomy (Helfaer et al., 1996; Sanders et al., 2006; Leong and Davis, 2007).
Preoperative sedation with benzodiazepines and postoperative treatment with opioids should be tailored to meet the needs of the patient. It would be prudent to titrate opioids, anticipating that the more severe the OSAS, the higher is the risk of opioid and benzodiazepine sensitivity. For patients with moderate or severe OSAS, many anesthesiologists prefer not to administer opioids until the patient has fully emerged from the anesthetic, has regained spontaneous ventilation, has been extubated, and is able to maintain an open airway with spontaneous ventilation. The patient’s opioid therapy may then be titrated carefully to achieve analgesia without respiratory compromise (Leong and Davis, 2007). During the immediate postoperative period, monitoring these patients in a postanesthetic care unit staffed with pediatric recovery room nurses allows a safe postoperative recovery with pain control.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree