FIGURE 39.1 Standard bag-valve-mask setup. Note that the bag is self-inflating, so it can be used with (usual) or without (in emergencies) an external gas supply. The “tail” of the bag serves as an oxygen reservoir.

FIGURE 39.2 A Mapleson D bag. Note that this is not a self-inflating bag, and hence must be used with an external gas source. The positioning of the fresh gas inlet—designating the Mapleson bag class—and the fresh gas flow impact the amount of rebreathing. It is possible, in an inadvertent situation, if the fresh gas runs out and the pressure regulating (“pop off”) valve is closed, to continuously rebreathe exhaled gas. This would ultimately result in injury or death.
| TABLE 39.1 Indications for Tracheal Intubation |
 |
Pediatric Considerations
Several anatomic differences exist between the adult and the pediatric airways. Pediatric patients have a relatively large head and flexible neck. Their occiput is larger by comparison and these differences make positioning a challenge forcing the head into a natural position of flexion. The air passages are physically smaller and shorter. However, the opening to the airway is higher, and more anterior, in children and varies with age. For example, in infants the opening is juxtaposed to the level of the first cervical vertebrae, by age 7, it has migrated inferiorly to the third cervical vertebrae and by adulthood somewhere about C5 or C6. The narrowest portion of the airway in a child occurs at the level of the cricoid cartilage, approximately located by the fourth cervical vertebrae, creating a funnel-shaped appearance to their airway. This finding differs from the narrowest part in an adult, the glottis aperture and vocal cords. Additionally, the pediatric vocal cords have a shorter distance from the carina, with the mainstem bronchus angulating symmetrically at the level of the carina at about 55 degrees. In adults, the right mainstem angulates at about 25 degrees and the left at about 45 degrees. The cupulae of the lungs are higher in the infant’s neck, increasing the risk of lung trauma. The tongue is relatively large for the mouth, the adenoids and tonsils are more apt to cause obstruction due their relatively large structure as well as function, and the epiglottis is longer, cephalad by comparison and more “floppy” than an adult’s causing greater hindrance. The glottis is typically slanted at a 40- to 50-degree angle, making intubation more difficult. Mucous membranes are softer, more pliable, and more fragile. These tissues readily become edematous when manipulated or an oversized ET is used. Finally, because the pediatric metabolic rate is much higher and oxygen consumption is much greater, these factors coupled with smaller functional residual capacity and highly compliant chest walls, the propensity for rapid desaturation exceeds the reserves of the adult patient (23).
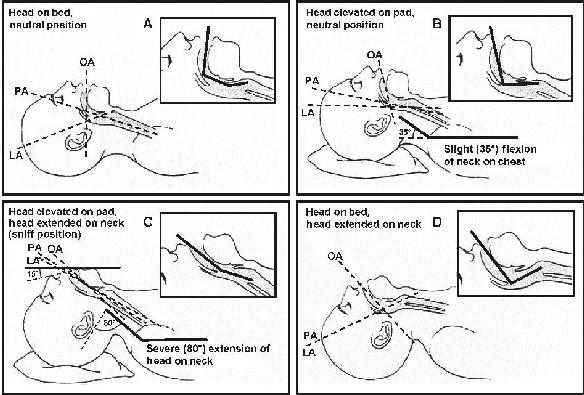
FIGURE 39.3 Demonstration of the “sniffing position” for optimal visualization of the glottic opening.

FIGURE 39.4 Laryngoscopic landmarks. Panel shows the cricoid cartilage.
EQUIPMENT FOR ACCESSING THE AIRWAY
Airway vigilance should be a goal of the critical care practitioner; thus, conventional and advanced airway rescue equipment must be immediately available during any attempts at airway management. Before attempting to intubate, all anticipated equipment and drugs must be prepared. Standard equipment for any translaryngeal, endotracheal intubation is not universal, in fact it is quite provider dependent. However, certain items have clearly demonstrated necessity and can be addressed in broad stroke. To avoid delay and minimize complications, it is imperative that all anticipated equipment and drugs must be available for the planned intubation technique (Table 39.2 and Fig. 39.5). This approach may best be facilitated by an organized “intubation box” containing conventional intubation equipment, with a selection of lubricants, local/topical anesthetics, intravenous induction agents, and medications to assist in treating peri-intubation hemodynamic alterations (heart rate, blood pressure). The box should have a visible lock with hand-breakable deterrent devices to reduce the problem of “missing” equipment. The wide spectrum of patient preparation for tracheal intubation ranges from an unconscious and paralyzed patient, to preparation with mild-to-moderate dosing of sedatives and analgesics, to the other extreme of topical anesthetics or no medication at all. Additionally, a difficult airway cart or bag with a variety of airway rescue devices—as well as a bronchoscope and/or fiberoptic laryngoscope—should be readily available (24,25). It is far better to have a limited assortment of airway devices with which personnel are familiar and competent to handle than to have an expensive, well-stocked cart containing a plethora of devices that the airway personnel have not practiced with nor have gained competence. A simple mnemonic to recall all of the necessary items required for intubation is S.O.A.P. M.E. (Suction, Oxygen source, Airway devices, Positioning and Pharmacy, Monitoring Equipment).
| TABLE 39.2 Standard Equipment and Drugs for Translaryngeal Intubation |
 |
Suction serves to facilitate visualization of the glottis by removing accumulated secretions such as saliva and blood. It also provides an essential measure of safety, in the event of regurgitation, against tracheal aspiration. Remember, no suction; no induction! An oxygen source can be a portable tank or wall-supplied. A method of delivery is also essential, at a minimum, a bag-valve-mask apparatus is needed to assist ventilation either noninvasively or following intubation. Airway equipment can be a highly variable list based upon provider preference and facility supply. As mentioned earlier, the emergency airway is neither the time nor the place to try an unfamiliar technique to secure the airway. There are, however, certain devices with which everyone working in critical care should be familiar including supraglottic devices, endotracheal intubation, video laryngoscopy (VL), and fiberoptic bronchoscopy.

FIGURE 39.5 Demonstration of the equipment and drugs that must be available for the planned intubation technique.
Esophagotracheal Combitube
The esophagotracheal airway (Combitube, ETC) (Fig. 39.6), recommended by the American Heart Association (AHA) advanced cardiovascular life support (ACLS) course and other national guidelines (24,26,27), is an advanced variant of the older esophageal obturator airway and the pharyngeal tracheal lumen airway (PTLA). The double-lumen system, with proximal and distal cuffs, allows ventilation and oxygenation in a majority of unconscious patients whether placed in the esophagus (95% of all insertions) or the trachea (28,29). Its proximal cuff is placed between the base of the tongue and the hard palate and the distal cuff within the trachea or upper esophagus (30,31). The ETC is inserted blindly, assisted by a jaw thrust or laryngoscopic assistance. Its role in emergency airway management is well recognized, and though less popular than the laryngeal mask airway (LMA), it may serve a vital role in offering airway rescue when laryngoscopy, bougie insertion, or LMA-assisted ventilation/intubation fails (32). A recent latex-free modification of the Combitube, the Easytuber (Teleflex Ruesch; www.teleflexmedical.com) has a shorter and thinner pharyngeal section, which allows the passage of a fiberscope via an opening of the pharyngeal lumen to inspect the trachea while ventilating (33).
Laryngeal Mask Airway
The most common supraglottic device in use today is the LMA. While typically used in elective surgical situations, its role in emergency airway management is unparalleled. As will be discussed later in the chapter in the section regarding difficult airway management, the LMA is the first rescue device employed in a “cannot ventilate” situation. The device is designed to create a seal in the hypopharynx while maintaining a conduit for ventilation. It does not provide a secure airway, but if inspiratory pressures remain low, it will reliably minimize abdominal insufflation. Newer modifications to the original design offer the ability to pass a suction catheter distal to the device without disturbing the seal in the hypopharynx.
Tracheal Intubation
Most commonly, orotracheal intubation is the preferred procedure to establish an airway because it usually can be performed more rapidly, offers a direct view of the glottis, has fewer bleeding complications, avoids nasal mucosal trauma, pressure necrosis, and the potential for sinus infection and allows a larger tracheal tube to be placed as compared to the nasal approach.

FIGURE 39.6 The esophagotracheal double-lumen airway, the Combitube.
Equipment
Laryngoscopy. A laryngoscope (Fig. 39.7) (video vs. conventional) is used to expose and visualize the glottis to facilitate passage of the tracheal tube. Unfortunately, proper skill and experience using this standard airway management technique varies widely among critical care practitioners. The utility of the laryngoscope under elective circumstances, with otherwise healthy surgical patients, is essentially limited to individuals with grade I or II views that can be easily intubated. Though a difficult view is mentioned by many as being uncommon, Kaplan et al. (17,34) documented a 14% incidence of grade III or IV views despite optimizing maneuvers such as the optimal external laryngeal manipulation (OELM) and the backward upward right pressure (BURP) technique (Fig. 39.8). These anatomic problems can be further complicated, as up to 33% of critically ill patients have a limited view with laryngoscopy (epiglottis only or no view at all) (35–37). These initial failures to intubate are why the critical care practitioner responsible for airway management must be familiar with alternatives to conventional DL. A review of the American Society of Anesthesiologist’s (ASA) Difficult Airway Algorithm will address this subject later.

FIGURE 39.7 Examples of fiberoptic laryngoscope handle and blades, in which the bulb is in the handle and the light is transmitted through fiberoptic bundles.

FIGURE 39.8 Diagrammatic representation of the optimal external laryngeal movement (OELM) and backward upward rightward pressure (BURP) maneuvers for optimal visualization of the glottis.

FIGURE 39.9 Various types of laryngoscope blades in common use.
Blades. Laryngoscope blades are of two principal kinds, curved (Macintosh type) and straight (typically, Miller type), varying in size for use in infants, children, or adults (Fig. 39.9). Many varieties of both the curved and straight blades have been redesigned in the hopes of augmenting visualization to facilitate passage of an ET. Innovations to improve laryngeal exposure include a hinged blade tip to augment epiglottic lifting during laryngoscopy (38), rigid fiberscopes, and video-assisted laryngoscopy. These innovations may, depending on the individual patient airway characteristics, offer an improved view of the glottis to improve the first-pass success rate, reduce intubation attempts, potentially reduce the time to intubation in the difficult airway, and potentially result in a reduction in esophageal intubation (EI) and other airway-related complications that are relatively commonplace with standard techniques (39–45).
Blade Use
The Curved Blade. Following opening of the mouth, either by the extraoral technique (finger pressing downward on chin) or the intraoral method (the finger scissor technique to spread the dentition), the laryngoscope blade is introduced at the right side of the mouth and advanced to the midline, displacing the tongue to the left. The epiglottis is seen at the base of the tongue and the tip of the blade inserted into the vallecula. If the oropharynx is dry, lubricating the blade is helpful; otherwise, suctioning out excessive secretions may assist greatly in visualizing airway structures. The laryngoscope blade should be lifted toward an imaginary point in the corner of the wall opposite the patient to avoid using the upper teeth as a fulcrum for the laryngoscope blade. Moreover, a forward and upward lift of the laryngoscope and blade at a 45-degree angle stretches the hyoepiglottic ligament, thus folding the epiglottis upward and further exposing the glottis. As a result, the larynx is suspended on the tip of the blade by the hyoid bone. The practitioner’s right hand, prior to picking up the ET, should be used to apply external pressure on the laryngeal cartilage (thyroid cartilage) to potentially afford better visualization of the glottis. OELM (see Fig. 39.8), as this maneuver is called, is optimized and turned over to an assistant who attempts to replicate the optimal position for the operator’s viewing. This description, while obviously optimal, is not always feasible.
With visualization of the glottic structures, the ET is passed to the right of the laryngoscope through the glottis into the trachea until the cuff passes 2 to 3 cm beyond the vocal cords. As described earlier, a Lehane–Cormack grade II or III airway may preclude easy placement of the tracheal tube. Thus, a blind guide underneath the epiglottis (tracheal tube introducer [TTI], bougie) or a rigid fiberoptic stylet may be incorporated to improve the insertion success rate.
The Straight Blade. Intubation with a straight blade involves the same maneuvers but with one major difference. The blade is slipped beneath the epiglottis and exposure of the larynx is accomplished by an upward and forward lift at a 45-degree angle toward the corner of the wall/ceiling interface opposite the patient. Again, leverage must not be applied against the upper teeth.
With either technique, the common causes of failure to intubate include inadequate position of the head, misplacement of the laryngoscope blade, inadequate muscle relaxation, insufficient depth of sedation/analgesia or general anesthesia, obscuring of the glottis by the tongue, and lack of familiarity with the anatomy, especially where pathologic changes are present. Inserting a laryngoscope blade too deeply, usually past the larynx and into the cricopharyngeal area, results in lifting of the entire larynx. If familiar landmarks are not appreciated, stop advancing the scope, withdraw the blade, and start over. If more than 30 seconds have passed or there is evidence that the oxygen saturation has dropped from the prelaryngoscopy level, bag-mask support to reoxygenate the patient is imperative. There is now evidence that repetitive laryngoscopies are not in the best interest of patient care and may place the patient at extreme risk for potentially life-threatening airway-related complications (35,36). Unless the first one to two laryngoscopy attempts were performed by less experienced members of the team, attempts at conventional laryngoscopy alone to intubate the trachea should be abandoned in favor of incorporating an airway adjunct to assist the clinician in hastening the process of gaining airway control (24,26,27,35,36,46,47).
Endotracheal Tubes. Most ETs are disposable and are made of clear, pliable polyvinylchloride, with little tendency to kink until they attain body temperature. Though the ETs mold to the contour of the upper airway and present a smooth interior, affording easy passage of suction catheters or a flexible bronchoscope, they may become encrusted with secretions, biofilm, and concretions that may decrease luminal patency and endanger patient care.
In adults, all commonly used ETs are of the cuffed variety. Many styles in use for critical care allow suctioning of subglottic secretions—the Hi–Lo Evacuation ET (Fig. 39.10). The ET cuff ensures a closed system, permitting control of ventilation and reducing the possibility of silent or active aspiration of oronasal secretions, vomitus or blood, although microaspiration is well recognized. Commonly, ET cuffs are the high volume–low pressure models that offer a broad contact with the tracheal wall and potentially limit ischemic damage to the mucosa. The tube size used depends upon the size of the patient (Table 39.3).
The primary reasons for tracheal intubation will vary by the patient’s pathophysiology, but also the physician’s judgment and experience in caring for the critically ill. The main goals of tracheal intubation include protecting the airway from contamination, providing positive pressure ventilation, providing a patent airway and permitting access to the tracheobronchial tree for suctioning, instillation of medications or diagnostic/therapeutic bronchoscopy. While the vast majority of tracheal intubations are via the oral route, the choice between the oral and the nasal—or the transcricoid/transtracheal route—will again be primarily determined by the patient’s physical status and airway anatomy, the expected duration of mechanical support and the judgment and skills of the practitioner.

FIGURE 39.10 The Mallinckrodt Hi–Lo Evacuation tube. While it comes in various sizes, it is not optimal for all patients. There is level 1 evidence that, with proper use, it decreases risk of ventilator-associated pneumonia. (Image courtesy of Medtronics).
Malleable Stylet. A well-lubricated malleable stylet (Fig. 39.11) is preferred by many to preform the ET into a shape that may expedite passage through the glottis. The stylet should be viewed as a guide, not a “spear,” and its tip should be safely inside the ET, never distal to the ET tip (48–51). It should not be used to force the ET into the airway or ram its way through the vocal cords when they are closed or otherwise inaccessible. A hockey stick–shaped configuration used by many can facilitate placement, but its angle must be appreciated cautiously. The curvature will often impede advancement into the airway since the ET tip may impinge on the anterior tracheal wall and the acute angulation of the stylet may impede its own removal from the ET (50–52). Ideally, the styleted ET tip should be placed at the entrance of the glottis, and then, with stylet removal, the ET will advance into the trachea less traumatically. Unfortunately, many practitioners unknowingly advance the styleted ET deep into the trachea without appreciating the potential damage the stylet-stiffened ET tip may cause to the tracheal wall.
| TABLE 39.3 Recommended Sizes for Endotracheal Tubes | |
 | |

FIGURE 39.11 Malleable stylet for use with insertion of an endotracheal tube.
POSITIONING THE PATIENT
One of the most important factors in improving the success rate of orotracheal intubation is positioning the patient properly (see Fig. 39.3). Classically, the sniffing position, namely cervical flexion combined with atlanto-occipital extension, will assist in improving the line of sight of the intubator. Bringing the three axes into alignment (oral, pharyngeal, and laryngeal) is commonly optimized by placing a firm towel or pillow beneath the head (providing mild cervical flexion) combined with physical backward movement of the head at the atlanto-occipital joint via manual extension. These maneuvers, when combined with oral laryngoscopy, will improve the “line of sight” for the provider to better visualize the laryngeal structures in most patients (53). Optimizing bed position is imperative, as is the angle at which the patient lies on the bed. The variety of mattress material (air, water, foam, gel) provides a challenge to the practitioner since these mattresses may worsen positioning characteristics in an emergency setting. Optimizing the position of the obese (Fig. 39.12A) patient is an absolute requirement to assist with spontaneous ventilation and mask ventilation; opening the mouth; gaining access to the neck for cricoid application, manipulation of laryngeal structures, or invasive procedures; improving the “line of sight” with laryngoscopy; and prolonging oxygen saturation after induction (54–58). A ramp is constructed with blankets, a preformed wedge or angulation of the mechanical bed to bring the ear and the sternal notch into alignment by ramping the patient’s head, shoulders, and upper torso, thus facilitating spontaneous ventilation, mask ventilation, and laryngoscopy. The extra time spent to properly position the patient is paramount to successful efforts (42,55,58).
MEDICATIONS
The pharynx, larynx, and trachea contain a rich network of sensory innervation, necessitating the use of anesthesia, analgesia, sedation, and sometimes muscular paralysis during intubation. Drugs commonly used are local anesthetics, sedative–hypnotics (sodium thiopental, propofol, etomidate), narcotics (fentanyl, morphine sulfate, hydromorphone, remifentanil), sedative-anxiolytics (benzodiazepine class—midazolam), muscle relaxants (depolarizing and nondepolarizing agents), and miscellaneous agents such as ketamine and dexmedetomidine. Critically ill patients often require only a fraction of the drug doses provided to their elective operating room counterparts. Careful intravenous titration may attenuate hemodynamic alterations, loss of consciousness, apnea, and aspiration. Controversy lies in whether or not to preserve spontaneous ventilation. Advocates for paralysis, the majority of which practice in the emergency department locale, cite a low rate of complications, and ease of intubation. Conversely, critical care databases suggest that emergency tracheal intubation is far from “safe” and devoid of complications whether or not paralyzing agents are administered (35,36,59–62). From a patient advocate standpoint, any practitioner who ablates the patient’s ability to spontaneously ventilate via neuromuscular blocking agents must be properly trained and experienced in basic and advanced airway management so that the depth of his or her ability to provide airway control lies well beyond simply conventional laryngoscopy and intubation (36).

FIGURE 39.12 Ramping of an obese patient’s torso to improve glottic visualization is noted on the left panel. The right panel shows the patient position without proper ramping.
Local Anesthetics
The use of local anesthetics is often overlooked in the intensive care unit (ICU) setting for a number of reasons:
- It is far easier to administer an intravenous agent than to take the time to anesthetize the airway with topical anesthetics or local nerve blocks.
- The urgency of the situation may preclude their timely use.
- The patient’s anatomic/physical characteristics may limit their effective application (poor or nonexistent landmarks, coagulopathy, excessively dry mucosa, excessive secretions, patient cooperation, physical or mechanical impediments).
- The lack of appreciation for their value in managing the airway.
Moreover, access to the proper local anesthetic agents and the accessories for their accurate delivery (nebulizer, atomizer, Krause forceps, cotton balls, Abraham laryngeal cannula, etc.) may be limited in the ICU setting unless they have been prepared and gathered in advance (difficult airway cart).
Aerosolized or nebulized 1% to 4% lidocaine can readily achieve nasopharyngeal and oropharyngeal anesthesia if the patient is cooperative and capable of deep inhalation. While this method is somewhat effective, it is less desirable due to its time-consuming application process and its limited success when compared to topically applied local anesthetics or local blocks. Transtracheal (cricothyroid membrane) instillation of 2 to 4 mL of 1% to 4% lidocaine with a 22- to 25-gauge needle causes sufficient coughing-induced reflex to afford ample distribution to anesthetize the subglottic and supraglottic regions plus the posterior pharynx in 90% of patients (63–65). Cocaine provides excellent conditions for facilitating intubation through the nasopharynx due to its outstanding topical anesthetic and mucosal and vascular shrinkage capabilities (64). However, in-hospital availability may limit its use in favor of phenylephrine or oxymetazoline combined with readily available local anesthetics. Lidocaine ointment applied to the base of the tongue with a tongue blade or similar device allows performance of DL in many patients. If time permits, nasal spraying with a vasoconstrictor followed by passing a progressively larger nasal airway coated with lidocaine provides exceptional coverage of the nasal cavity in preparing for a nasal intubation. Instillation of liquid lidocaine via the in situ nasal trumpet offers an excellent conduit to distribute additional topical anesthetic to the orohypopharynx. It is best performed in the sitting-up position to enhance coverage of the airway structures.
Narcotics
Narcotics such as morphine, hydromorphone, fentanyl, and remifentanil reduce pain perception making intubation less stressful. In addition, they have some sedative effect, suppress cough, and relieve dyspnea (66,67). Fentanyl and the ultrashort-acting remifentanil have a more rapid-onset and shorter duration of action than the conventional narcotics used in the ICU setting for analgesia (68–70). Morphine may lead to histamine release and its potential sequelae. All narcotics cause respiratory depression, the newer synthetic narcotics may also lead to muscular chest wall rigidity that may hamper ventilation, and may contribute to episodes of bradycardia. Their analgesic and antitussive qualities are extremely valuable especially given the ready availability of a method to reverse excessive narcotization.
Benzodiazepines
Benzodiazepines such as lorazepam and midazolam have excellent amnestic and sedative properties (71). Diazepam has seen its use decline markedly due to its less favorable distribution and clearance characteristics. This drug class does not provide analgesia and may be combined with an analgesic agent during intubation, especially if an awake or semiconscious state with maintenance of spontaneous ventilation is the goal. Midazolam is preferred to lorazepam use for intubation if possible, due to lorazepam’s slower pharmacodynamic onset (2 to 6 minutes). Hypotension may occur in hypovolemic patients and benzodiazepines potentiate narcotic-induced respiratory depression.
Propofol
Propofol is extremely useful during intubation, especially if slowly titrated to a desired effect rather than simply administering a one-time bolus). If done correctly, maintenance of spontaneous ventilation is possible, while still achieving adequate levels of sedation. After intravenous administration via a peripheral IV (1 to 3 mg/kg ideal body weight [IBW]), unconsciousness occurs within 30 to 60 seconds. Awakening is observed in 4 to 6 minutes with a lower lingering level of sedation compared to other induction agents (72,73,75). Side effects include pain on injection, involuntary muscle movement, coughing, and hiccups. Hypotension, cardiovascular collapse and, rarely, bradycardia may complicate its use, especially if administered in rapid single-bolus dosing in the critically ill patient with relative or absolute hypovolemia, a systemic capillary leak syndrome, or pre-existing vasodilatation (e.g., sepsis, systemic inflammatory response syndrome [SIRS]). It is important to consider that propofol has no analgesic properties and may benefit from adjunctive agents to provide pain relief.
Ketamine
Ketamine, a phencyclidine derivative, provides profound analgesia, amnesia, and dissociative anesthesia (76,77). Airway reflexes and spontaneous ventilation are often, but not always, preserved. Ketamine has a rapid-onset and relatively short duration of action. Its profile is unique: it is a myocardial depressant, but this is often countered by its sympathomimetic properties, thus leading to hypertension and tachycardia in many patients. However, in patients who are catecholamine depleted, the sympathomimetic effects pale in comparison to its depressant side effects leading to hypotension. Though it also offers favorable bronchodilatory properties, it does cause bronchorrhea, salivation, and a high incidence of dreams, hallucinations, and emergence delirium, the latter of which can be ameliorated with concomitant benzodiazepine use (76,77).
Etomidate
Etomidate was considered by many to be the preferred induction agent in the critically ill patient due to its favorable hemodynamic profile, as compared to the other available induction agents (78). Recently, the drug has come under intense scrutiny due to newer research associating it with higher morbidity and mortality in sepsis (79,80). The hemodynamic stabilization offered by etomidate, however, should not be considered a panacea since it too may lead to hemodynamic deterioration (81,82). Its role as a single-dose induction agent is in question due to its transient depression of the adrenal axis. Once regarded as a minor concern, this adrenal suppression has been shown to affect the outcomes of the critically ill. Some have expressed caution with etomidate’s use as a single-dose induction agent, especially in the septic or trauma populations. Although recent retrospective information gathered from an ICU database in patients with sepsis who received etomidate demonstrates no increased mortality from single dose usage (74), a variety of opinions exist, ranging from an opinion that etomidate should be avoided completely, to its avoidance in select populations such as the septic population, to its use with concurrent steroid replacement therapy for at least 24 hours (81–83). If administration of etomidate is necessary, its use should be discussed with the ICU team should future complications arise.
Dexmedetomidine
Dexmedetomidine is an intravenous ultrashort-acting α2-agonist that provides analgesia and mild-to-moderate sedation with minimal respiratory depression while affording tolerance of “awake” fiberoptic and conventional tracheal intubation (84–86). An initial bolus dose of 1 mcg/kg, followed by a continuous infusion of 0.2 to 0.7 μg/kg/min should be adequate in most cases. Its most noted side effects are hypotension and bradycardia. While a useful drug, it can be cost prohibitive, particularly when compared to propofol (87,88). Its best used as part of a balanced multimodal technique to obtain conditions fit for intubation, but is an excellent long-term sedative alone.
Barbiturates
Sodium thiopental, an ultra-short–acting barbiturate, decreases the level of consciousness and provides amnesia without analgesia after an intubation dose of 4 to 7 mg/kg IBW dose over 20 to 50 seconds (administered via a peripheral IV) in the otherwise healthy patient. Its short duration of action (5 to 10 minutes) makes it ideal for short procedures such as intubation. Thiopental has an excellent cerebral metabolic profile in regards to lowering cerebral metabolic rate while maintaining cerebral blood flow as long as systemic blood pressure is maintained within an adequate range. However, thiopental may lead to hypotension in critically ill patients due to its vasodilatation properties, especially in the face of hypovolemia (63). Though inexpensive, its use in the operating room has declined in favor of propofol. In the ICU setting, reducing the dose of thiopental to 1 to 2 mg/kg IBW is very useful for preparing the patient for tracheal intubation with or without a muscle relaxant. Unfortunately, due to legal import restrictions regarding the drug’s use in capital punishment, it is no longer available in the United States.
Muscle Relaxants
The administration of a sedative–hypnotic agent with a rapid-acting muscle relaxant, typically succinylcholine, as the standard technique for tracheal intubation is often cited as improving intubation conditions and leading to fewer complications (59). However, most ICU intubations can be accomplished without their use. Though this recommendation has merit, its ubiquitous acceptance can get an inexperienced laryngoscopist in to trouble when unable to intubate or ventilate (35,36,60,61). It is our opinion that any clinician who administers drugs which induce anesthesia or cause paralysis, thus rendering the patient entirely dependent on the airway management team, should have a contingency strategy complete with the equipment to deploy such a tactic (35,36,60).
Indications for muscle relaxants include agitation or lack of cooperation not related to inadequate sedation, increased muscle tone (seizures, tetanus, and neurologic diseases), avoidance of intracranial hypertension, limiting patient movement due to comorbidity, and the need for rapid tracheal intubation.
Neuromuscular blocking agents may cause depolarization of the motor end plate (succinylcholine) or prevent depolarization (cisatracurium, vecuronium, rocuronium). Succinylcholine has a rapid-onset and short duration of action, making it useful in the critical care setting; however, it may raise serum potassium levels by 0.5 to 1.0 mEq/L. It is contraindicated in chronically immobilized patients, skeletal myopathies, hyperkalemia, burns, or recent or long-term neurologic deficits (89–91). Other side effects are elevation of intragastric and intraocular pressures, muscle fasciculation, myalgias, malignant hyperthermia (it is contraindicated if there is a history of MH), bradyarrhythmias, and myoglobinuria. Depending on the initial dose—our recommendation is 0.25 to 1 mg/kg IBW—and systemic conditions, succinylcholine has a relatively short duration of 3 to 10 minutes. In patients with pseudocholinesterase deficiency, paralysis may last as long as 24 hours. In the event of failed intubation, duration of action is irrelevant and emergency actions should be employed immediately (92).
Nondepolarizing muscle relaxants have a longer time to onset and duration of action as compared to succinylcholine. Rocuronium (typical operating room dose, 0.6 mg/kg) can mimic the time of onset of succinylcholine if dosed accordingly (1.2 mg/kg), but the increased dosage requirements extend the duration of drug action and increased cost. While the rapidity of onset and shorter duration of action have made Rocuronium a commonly chosen agent, comorbidities must be considered prior to administration. Rocuronium can cause histamine release in approximately 1% of patients and may cause anaphylaxis (93,94). Additionally, due to its hepatic metabolism, duration of action may be prolonged by 1.5 times in patients with liver failure. Vecuronium has a slightly slower onset of action, approximately 3 to 5 minutes with a regular–rapid intubating dose of 0.1 to 0.3 mg/kg. It does not cause histamine release. Cisatracurium is a unique muscle relaxant because of its metabolism. The standard intubating dose is 0.1 to 0.2 mg/kg providing an “intermediate” onset and duration of action compared to the other two agents. The advantages of cisatracurium are limited hemodynamic effects, no histamine release, and consistent pharmacokinetics despite renal or hepatic failure due to Hoffmann elimination of the drug (94).
MONITORING EQUIPMENT
Monitoring equipment for intubation falls into three main categories: hemodynamics, oxygenation, and ventilation. Heart rate, electrocardiogram, and blood pressure should all be monitored routinely because not only the medications previously discussed affect hemodynamics, but so does the procedure itself, quite profoundly in many cases, in fact. Adequate oxygenation can be assessed by pulse oximetry with some reliability. There are instances where changes in oximetric readings will lag behind true changes in oxygenation. Additionally, there may be conditions which cause false readings, such carbon monoxide toxicity or methemoglobinemia. Adequate preoxygenation, or more appropriately denitrogenation of the functional residual capacity, can be assessed by measuring end-tidal oxygen levels; however, outside of the operating room, this technology is rarely seen. In most cases, an appropriate level of preparation can be assumed with 3 to 5 minutes of preoxygenation with 100% oxygen.
Adequate ventilation can be determined by measuring the presence of end-tidal carbon dioxide. This task can be accomplished by capnography. The identification of exhaled CO2 via disposable colorimetric devices or capnography should be considered an accepted standard of practice for elective as well as out-of-the-operating-room intubation (24,51,95). Considered “almost fail-safe,” these methods may fail due to a variety of causes. Specifically, the disposable colorimetric devices cannot determine CO2 with poor cardiac output. A color change may partially or completely fail due to expiration or previous usage or, most commonly, misinterpretation may occur due to soiling from secretions, pulmonary edema fluid, or blood. Conversely, capnography may fail due to temperature variations (outside, helicopter rescue), soiling of the detector, battery or electrical failure, equipment failure due to age, missing accessories, or lack of maintenance.
AIRWAY ACCESS TECHNIQUES
General Indications and Contraindications
The oral approach is the standard method for tracheal intubation today. The indications are numerous and it may be the best to focus on the contraindications. The oral route would not be a reasonable choice when there is limited access to the oral cavity due to trauma, edema, or anatomic difficulties. Though nasal intubations were a mainstay in earlier decades, the oral approach has displaced it due to the popularity of the “rapid sequence intubation” (RSI) and the better appreciation of the potential detriments of long-term nasal intubation. As such, a surgical approach via the cricothyroid membrane or a formal tracheostomy has typically become the alternative choice in a critical care setting.
Orotracheal Intubation
Adherence to the aforementioned checklist of supplies is crucial. While these items are assembled, preoxygenation should be accomplished. “Ramping” the obese patient and adjusting the bed height and angulation should be carried out (see Fig. 39.12A); how not to position is noted in Figure 39.12B. A rapid medical–surgical history should also be obtained, including a review of previous intubation attempts and an airway examination (24,51). Intravenous access is essential. As in any emergency situation, a clear delineation of participant roles will eliminate confusion.
A tube of appropriate diameter and length should be selected and, though gender is an important factor in size selection, patient height is equally important as there is a linear relationship between the latter and glottic size. Typically, the choice in a woman would be a 7- to 8-mm ET, and in males an 8- to 9-mm ET. Smaller diameter ETs should be available for any eventuality. The ET should be examined for patency and cuff integrity. The 15-mm proximal adapter, universal for all airway device connectors, should fit snugly and the ET kept in its sterile wrapper and not handled until insertion. The ET may be placed in warm water to soften the PVC tubing, improving passage of the ET over a stylet, tracheal introducing catheter (bougie), fiberoptic bronchoscope, or an ET exchange catheter.
Patient preparation for tracheal intubation may range from little to no medication to the other extreme of unconsciousness with muscle relaxation (59,96,97). Airway risk assessment must be weighed against the preservation of spontaneous ventilation and the depth of anesthesia necessary to tolerate intubation (24,26,27). The pharmacodynamic effects following administration via an IV site will depend on the IV location and patency, catheter diameter and length, IV flow rate, and the patient’s cardiac output. Central IV access may speed time to onset and potentially deliver a more concentrated medication bolus compared to a peripheral IV site.
The practitioner has several choices for patient preparation: awake with no medication; awake with topical anesthesia or local nerve blocks, with or without light sedation; sedation/analgesia only with the option of neuromuscular blocker use; and a set induction regimen for an RSI (35,36,42,59,96,97).
Awake Intubation
Awake intubation techniques require experience, may comprise either oral or nasal routes, and usually require topically applied local anesthetics (Fig. 39.13) or local nerve blocks with or without sedation (18,98). The advantage of this technique is maintenance of spontaneous ventilation during emergency airway management by avoiding excessive sedative–hypnotic agents and/or muscle relaxants (99). Awake intubation techniques have been largely supplanted by induction of unconsciousness or deep sedation with or without muscle relaxation (99,100). Though the “awake intubation” is an extremely useful approach, its reduced utilization may complicate patient care due to poor technique, inadequate topical anesthesia, ineffective local nerve blockade, and the lack of judicious or creative sedative and analgesic schemes.

FIGURE 39.13 Equipment used to topicalize the airway prior to instrumentation: tongue blade with lidocaine jelly, nebulizer with 4% lidocaine, and nasal dilators of various sizes.
Awake intubation may benefit from the addition of a narcotic agent by providing analgesia, antitussive action, and more stable hemodynamics. Many reserve an awake approach for the known or suspected difficult airway to avoid “burning any bridges.” However, an awake approach neither guarantees successful intubation, nor is it devoid of morbidity or mortality (101–103).
Following proper preparation, unless the patient is unconscious or has markedly depressed mental status, the “awake-look” technique incorporates conventional laryngoscopy to evaluate the patient’s airway to gauge the feasibility and ease of intubation (51); explanation to the patient (if applicable) is imperative for cooperation. If viewing the airway structures during an “awake look” proves fruitful, intubation should be performed during the same laryngoscopic attempt either directly—grade I or II view—or by bougie assistance or other means—grade I, II, or III (104,105). Many “awake-look” procedures that yield a reasonable view, but in which intubation is not performed, are followed by anesthetic induction with the potential for a worse view due to airway tissue collapse and obstruction by redundant tissue due to loss of pharyngeal tone. Too often, patient comfort is placed above patient safety. The critically ill patient is often tolerant of bougie-assisted intubation (Fig. 39.14), supraglottic airway (SGA) placement (e.g., LMA) (Fig. 39.15), or the placement of specialty airway devices such as the fiberoptic bronchoscope (FOB) following topical anesthetic application, local nerve blocks, or sedation (18,106–109).
Sedated to Asleep Techniques
Titration of medication to provide amnesia, analgesia, anxiolysis, sedation, or a combination of these desirable effects with the goal of providing comfort while preserving spontaneous ventilation is possible (35,110). Muscle relaxants may be added as an option if pharmacologic attempts to render the patient accepting of airway manipulation prove suboptimal or unsatisfactory. Sedation and amnesia are mandatory when paralysis is induced (35,61,99,100). Breath-holding, glottic closure, laryngospasm, swallowing, biting, jaw clenching, and gagging may contribute adversely to the intubation process, but most of these are overcome with patience (24,26,27).
Rapid Sequence Intubation
RSI refers to the administration of an induction agent followed by a neuromuscular blocking agent, to reduce the time to onset of unconsciousness and muscle paralysis based on a concern for aspiration. By minimizing the time the airway is unprotected, the risk of aspiration theoretically should be reduced. Preoxygenation is paramount in RSI since no active oxygenation and ventilation efforts are done, thus, hypothetically, avoiding esophagogastric insufflation (59,111,112). Cricoid pressure is applied, in theory, to reduce the risk of passive regurgitation of any stomach contents (22). These practices during an RSI may not always be practical since patients can desaturate during the apneic phase of the RSI, particularly in obesity, pregnancy, with suboptimal preoxygenation or in the presence of cardiopulmonary pathology.

FIGURE 39.14 Array of tracheal “bougies” used to access the airway in difficult situations.
If necessary, bag-mask support should be delivered despite the concern about esophagogastric insufflation and subsequent regurgitation/aspiration. Additionally, the application of cricoid pressure—both quantitative and qualitative—is so variable that concerns with its ubiquitous use and overall effectiveness have been raised (113–116). Cricoid pressure can improve or worsen the laryngoscopic view, and may impede mask ventilation; hence, adjustment or release of cricoid pressure should be considered in these circumstances. Further, cricoid pressure may alter the ability to place accessory devices, such as the LMA and impede fiberoptic viewing (117–120). Despite the potential limitations of cricoid pressure, no desaturating patient should have assistive ventilation withheld.
When performing an RSI, a preplanned strategy to assist the patient must be in place prior to induction. The development of such strategies during a crisis is difficult, often short-sighted, incomplete, and may be harmful to patient care. Education, training, and immediate access to airway rescue equipment that the practitioner can competently incorporate in an airway crisis is a goal worthy of expanded effort, time, and finances (24,26,27,35,51,59,61). Proponents of rapidly controlling the airway using RSI cite a reduction in the risk of aspiration as a main thrust for this technique. Moreover, RSI is said to be associated with a lower incidence of complications and higher first-pass intubation success rate as compared to a “sedation only” method (59,61,111,112). Though there is little argument that many intubations may be made easier by the administration of a muscle relaxant, selective use based on the patient evaluation and clinical circumstances is the best option (24,26,27,35–37,51,62,99,100).
Nasotracheal Intubation
Nasotracheal intubation is still commonly used in oral and maxillofacial operative interventions, but less commonly in emergency situations outside the operating room. Nasotracheal intubation is an alternative to the oral route for patients with trismus, mandibular fracture, or angioedema (121). It is also a useful approach for the spontaneously breathing patient who cannot lie supine or in the presence of excessive secretions. The presence of midfacial or posterior fossa trauma and coagulopathy are absolute contraindications to this technique. It is also contraindicated in the presence of acute sinusitis or mastoiditis. Additionally, as the nares dictate a smaller-diameter tracheal tube, and a smaller ET is necessary, the length of the tracheal tube is shortened; hence, the length must be considered to ensure entrance into the trachea (122–124).

FIGURE 39.15 Laryngeal mask airways for emergent/difficult intubation. A: The intubating laryngeal mask airway (LMA). B: Various sized LMAs for patients of different sizes and ages.
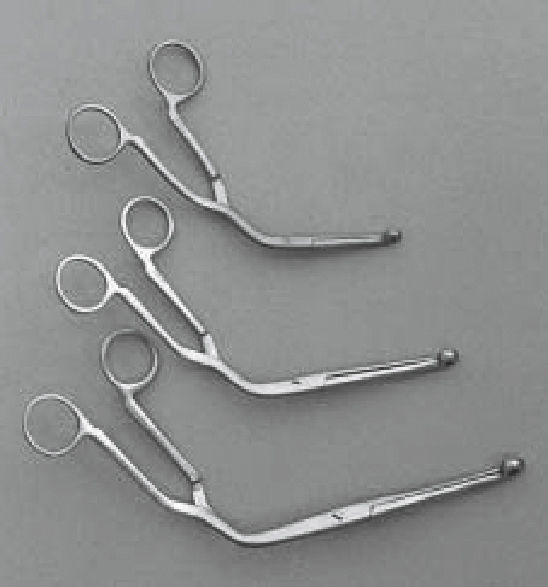
FIGURE 39.16 Magill forceps for manipulating the endotracheal tube into the glottis. These come in several sizes.
The method of intubation via the nasal approach is variable. It may be placed blindly during spontaneous ventilation, combined with oral laryngoscopic assistance to aid with ET advancement utilizing Magill forceps (Fig. 39.16), use indirect visualization through the nares via an optical stylet (Fig. 39.17), flexible (Fig. 39.18) or rigid FOB (Fig. 39.19), or incorporate a lighted stylet (Fig. 39.20) for transillumination of the laryngeal structures (43,125,126).
Technique
Pretreatment of the mucosa of both nostrils with a solution of 0.1% phenylephrine or decongestant spray such as oxymetazoline for 3 to 10 minutes is primary. Placement of cotton pledgets soaked in a mixture of vasoconstrictor agent and local anesthetic is equally effective. This action will minimize bleeding by shrinking vascularity. Vasoconstriction is followed by dilation, utilizing progressively larger nasal trumpets lubricated with 2% lidocaine jelly (see Fig. 39.13). The method is relatively expedient. Supplemental oxygen may be provided by a nasal cannula placed between the lips or via a face mask. The patient is best intubated with spontaneous ventilation maintained, yet incremental sedation may be provided to optimize patient comfort. Sitting upright has the advantage of maximizing the oropharyngeal diameter (122,127).

FIGURE 39.17 An optical stylet, allowing visualization of the glottis as the endotracheal tube is advanced.
Orientation of the tracheal tube bevel is important for patient comfort and to reduce the risk of epistaxis and tearing or dislocation of the nasal turbinates (128,129). On either side of the nose, the bevel should face the turbinate (away from the septum). Warming the tracheal tube combined with generous lubrication will assist rotation and advancement while providing a soft and pliable airway to reduce injury to the nasal mucosa or turbinates. Tube advancement should be slow and gentle, with rotation when resistance is encountered. Other options to resistance include sitting the patient upright, flexing the head forward on the neck and manually pulling the larynx anteriorly. Conversely, if advancement is met with posterior displacement into the esophagus, sitting the patient upright, extending the head on the neck, and applying posterior-directed pressure on the cricothyroid complex may assist in intubation. Often, one can then feel the inflation of the tracheal cuff below the larynx and above the manubrium (130). Confirmation with end-tidal CO2 measurement or fiberoptic viewing of subglottic anatomy is imperative.
Nasotracheal intubation in most elective situations is accomplished with transoral manipulation using the Magill forceps or with fiberoptic assistance. Application of a specially designed airway “whistle” that assists the clinician with spontaneous ventilation intubation may be advantageous when blind placement is attempted (131). Should either the blind or transoral approach cause trauma, fiberoptic assistance may be limited if secretion control is poor or bleeding is present. Fiberoptic bronchoscopy is an excellent choice for the primary nasal approach. Ideally, the provider should be standing in front or to the side of the patient as opposed to “over the top” or above the patient (132,133). Advancement of the ET into the glottis may be impeded by hang-up on the laryngeal structures: a vocal cord, the posterior glottis, or, typically, the right arytenoid cartilage. When resistance is met, a helpful tip is as follows: withdrawing the tube 1 to 2 cm, rotate the tube counterclockwise 90 degrees, then advancing with the bevel facing posteriorly (134,135). Matching the tracheal tube to the fiberscope to minimize the gap between the internal diameter of the tube and the scope may also improve advancement (134). Tracheal confirmation and tip positioning are added advantages to fiberoptic-assisted intubation.
Complications of Nasal Intubation
A nasally placed ET has the advantage of stability, but has lost favor due to limited ET size, the potential for epistaxis, a risk for sinus obstruction, ulceration, and infection beyond 48 hours and nasal mucosal damage. Avulsion of the turbinate bone when the ET engages the anterior edge of the middle turbinate’s lateral attachment is also possible (122–124,136,137–139).

FIGURE 39.18 A fiberoptic bronchoscope with associated cart as used at Shands Hospital at the University of Florida.
INTUBATION ADJUNCTS
Tracheal Tube Introducer or Bougie
The TTI (or bougie) (see Fig. 39.14) has earned a position in anesthesia care as an effective airway adjunct by assisting navigation of the ET into the trachea when anatomic constraints limit the view of the glottic opening (140). A grade II (arytenoids and posterior cords only) or grade III laryngeal view (epiglottis only) is ideal for bougie-assisted intubation (105,141). The TTI is listed as a rescue option in national guidelines and should be included in a difficult airway cart or portable bag (24–27). The advantages of the bougie include low cost, no power supply, portability, a rapid learning curve, minimal set-up time, and a relatively high success rate. Its immediate use reduces intubation-related complications (105,141). Placement involves passing it underneath the epiglottis with further navigation through the glottis to a depth of 20 to 24 cm. Potential tactile feedback can occur as the angulated tip bounces over the cartilaginous anterior trachea rings. The tracheal ring “clicks” may not be appreciated in all cases. Further gentle advancement to 28 to 34 cm leads to the “hang-up test” or Cheney test. This maneuver is useful not only for bougie-assisted intubation itself, but also when ET verification maneuvers and devices are imprecise or confusing. Passing the ET is assisted by laryngoscopy to clear the airway of obstacles, lubricating the ET, and counterclockwise rotation to limit arytenoid hang-up of the ET tip. The bougie’s role in difficult airway management is underappreciated and given its potentially prominent role as a simple airway tool, more attention to its position in an airway management strategy is warranted (46,47,142).

FIGURE 39.19 A rigid bronchoscope.
Indirect Visualization of the Airway
Fiberoptic Bronchoscopy
Flexible bronchoscopy is the gold standard in indirect visualization of the airway. It is the critically ill patient who certainly would benefit from improving the “line of sight,” a straight line from the operator’s eyes to the level of the glottic opening (34,37,45,143). Being able to see “around the corner” is immensely important when one’s goal is to minimize intubation attempts and hasten the time to securing the airway (39,42–44,52). Its role in the critically ill is broad and serves many life-saving roles, both diagnostic and therapeutic. Flexible bronchoscopy does require expertise and patience and may be limited by secretions and edema (132). Fiberoptic intubation best serves the critically ill patient as a first-line technique (132). This statement is in opposition to a rescue technique (Table 39.4) (20,47,143,144), where edema, secretions, and bleeding often complicate visualization of the airway following multiple failed conventional attempts.

FIGURE 39.20 A lighted stylet (Lightwand) for blind insertion of an endotracheal tube. Utilization of this technique requires significant practice.
| TABLE 39.4 Clinical Uses of Fiberoptic Bronchoscopy in the Intensive Care Unit |
 |
An external monitor to display the fiberoptic view (see Fig. 39.18) to the airway team is an excellent teaching modality and it allows input by other team members to optimize positioning and hasten the intubation process (132,145). Fiberoptic intubation effectiveness is reduced by inadequate patient preparation (e.g., topical local anesthesia application when mucosal desiccation or excessive secretions are present, excessive sedation in an attempt to counter poorly functioning topical anesthesia coverage or inadequate local anesthesia blocks). An inexperienced practitioner, one of the prime reasons for failure or suboptimal or no assistance (hence the inability to provide adequate jaw thrust or lingual retraction); improper choice of equipment (using a pediatric-sized bronchoscope to place a 9.0 ET); and improper positioning (using the supine approach in a morbidly obese patient) all will impact negatively on success. An awake technique chosen may further contribute to failure and frustration. Inadequate patient preparation with medication (e.g., too light sedation leading to discomfort or an uncooperative patient, or excessive sedation leading to hypoventilation, airway obstruction, or excessive coughing or procedural pain due to lack of narcotic administration) will place an undue and likely uncorrectable burden on the fiberoptic technique.
Successful fiberoptic intubation is dependent on a wide range of factors, a cooperative patient, bronchoscope defogging, adequate lubrication, experience, and good judgment in the approach (e.g., not performing a nasal approach in the face of a coagulopathy or nasofacial abnormalities, excessive bleeding or uncontrollable secretions) each being performed in a timely manner (Table 39.5). Any single factor that is neglected or improperly executed may hamper the fiberoptic effort.
Video Laryngoscopy and Rigid Fiberscopes
To overcome the difficulty of “seeing around the corner” (under the epiglottis, into the hypopharynx) various advancements have been made to the standard laryngoscope. Though a difficult-to-visualize glottis is said to be uncommon, Kaplan et al. (146) report that DL in a large cohort of elective general anesthesia patients had a Lehane–Cormack views of III or IV in 14% despite maneuvers to optimize viewing with a curved laryngoscope blade (34,147). The incidence of a grade III/IV view in the emergency intubation population is quoted to be more than double this rate; hence the need to improve visualization capabilities “around the corner” (35,148).
| TABLE 39.5 Keys to Fiberoptic Intubation Success |
 |
Devices such as the Bullard scope (26,41,146) (Fig. 39.21), the Wu scope (19,149,150), and the Upsher-scope rigid fiberscope (150) provide an indirect “around the corner” look (15,39,40). These devices should be considered first-generation variations, and while still available, are seldom used in deference to more modern video laryngoscopes.
The Macintosh (curved) video laryngoscope (Karl Storz endoscopy) was developed and produced by modifying a standard laryngoscope to contain a small video camera (34,151). While it adds greater viewing options, it seldom improves a view. Currently, improvements in video screen resolution, portable power sources, and the refinement in optics have afforded a new class of airway devices to assist in management of the difficult airway in the operating room and remote floor locations (19,43,146,152). The newest video laryngoscopes are variations on the curved blade with a more acute, approximately 60-degree tip deflection. The GlideScope and McGrath scope are the most common of these devices and very well may become the standard method for intubation (153,154). Though glottis visualization markedly improves in most cases, a principal observation to appreciate is that these instruments do not perform intubation of the trachea. Visualization of structures with failure to intubate is more uncommon (<4%) (152,155), but still possible and may require further adjuncts for success (156). Despite its reproducibility of success, a recent trauma analysis comparing to DL to VL, shows VL actually took longer to accomplish and had no impact on outcome (157).

FIGURE 39.21 Bullard intubating laryngoscope.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








